Abstract
Introduction
Acute myeloid leukemia (AML) has poor outcomes despite long term advancement in its diagnosis and treatment. The introduction of molecular genetic markers in AML has produced a major effect on therapeutic approach used in these patients. The genetic aberration in the Nucleophosmin (NPM1) gene is the most common mutation found in AML and is associated with "favorable" prognosis. Allogenic hematopoietic stem cell transplant (Allo-HSCT) is potentially a curative treatment option. Herein, we present a systematic review and meta-analysis with the aim of exploring outcomes with allo-HSCT in NPM1-mutated AML.
Methods
A detailed literature search was performed for the systematic review and meta-analysis in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. A population, intervention, comparison, and outcome table were developed, and three electronic databases (PubMed, Cochrane Register of Controlled Trials, and Clinical trials.gov) were searched using MeSH terms and keywords for "Transplantation Hematopoietic Stem Cell" AND "Nucleophosmine Protein, Human" AND "Acute Myeloid Leukemia". A total of 137 articles were screened and original studies reporting patients with AML having NPM1 mutation who underwent Allo-HSCT were included while reviews, duplicate, and non-relevant articles were excluded. A total of twelve studies were included. Quality evaluation was done using the NIH quality assessment tool. The inter-study variance was calculated using Der Simonian-Laird Estimator. The pooled analysis was conducted using the 'meta' package by Schwarzer et al. in the R programming language (version 4.16-2). A random-effects model was used to estimate the proportions with 95% confidence intervals (CI).
Results
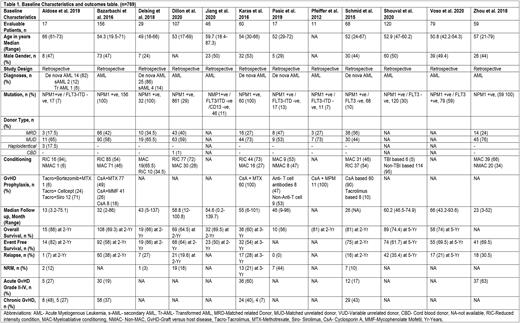
A total of 769 patients from twelve studies were included for this systematic review and meta-analysis. The median age of patients was 53 (21-79) years and 46.5% (303/651) were males as reported by ten studies. The donor type was matched related donor (MRD) 38%, matched unrelated donor (MUD) 61%, haploidentical 0.6%, and cord blood donor (CBD) 0.2%. The median follow-up was 46 (2-96) months. At a median follow-up of 2 (2-5) years, the pooled overall survival (OS) was 71% (95 % CI 0.66-0.73, p= 0.15, I2=31%, n= 710) while the pooled event free survival (EFS) and pooled relapse rate (RR) was 64% (95 % CI 0.59-0.68, p= 0.06, I2=45%, n= 741) and 25% (95 % CI 0.19-0.31, p <0.01, I2=70%, n= 695), respectively. The pooled incidence of acute graft versus host disease (GvHD) was 39% (95 % CI 0.16-0.65, p<0.01, I2=96%, n= 380) and the pooled incidence of chronic GvHD was 46% (95 % CI 0.35-0.58, p= 0.02, I2=71%, n= 301). The pooled incidence of non-relapse mortality (NRM) was 15% (95 % CI 0.08-0.23, p= 0.02, I2=63%, n= 298) at a median follow-up of 2 (2-5) years.
Conclusion
NPMI-mutated AML has favorable outcomes with allogeneic hematopoietic stem cell transplant and has good overall and event free survival with lower incidence of relapse and non-relapse mortality. Further randomized clinical trials are needed to consolidate these findings.
Disclosures
Abhyankar:Incyte: Consultancy, Research Funding, Speakers Bureau; Therakos: Consultancy, Research Funding, Speakers Bureau. McGuirk:Magenta Therapeutics: Consultancy, Honoraria, Research Funding; Allovir: Consultancy, Honoraria, Research Funding, Speakers Bureau; Juno Therapeutics: Consultancy, Honoraria, Research Funding; In8bio, Inc.: Other: IIT Clinical Trial; BMS: Consultancy, Honoraria, Speakers Bureau; Novartis: Consultancy, Honoraria; Kite, a Gilead Company: Consultancy, Honoraria, Research Funding, Speakers Bureau; Nextar: Consultancy, Honoraria; Orca Bio: Research Funding; CRISPR Therapeutics: Consultancy; Sana: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.